Blog Details
Blog Details
How to Choose a Biostatistics Partner for Oncology Programs
How to Choose a Biostatistics Partner for Oncology Programs
This Article — 5-Minute Read
This Article — 5-Minute Read


Oncology accounts for nearly half of all pharmaceutical R&D spending, yet Phase III oncology trials still fail at a staggering rate of roughly 65%. The uncomfortable truth is that most of these late stage failures have nothing to do with flawed biology. They stem from statistical design errors, inadequate powering, poorly defined endpoints, and an inability to navigate shifting regulatory expectations.
For early and mid stage oncology biotechs, selecting a biostatistics partner is not a procurement exercise. It is a strategic decision that directly dictates whether your clinical program survives.
This guide strips away the marketing fluff to cover what you actually need to know: the engagement models available, the real financial costs, the interview questions that separate true experts from pretenders, and the regulatory landmines that sink FDA applications.
The Four Models for Oncology Biostatistics Support
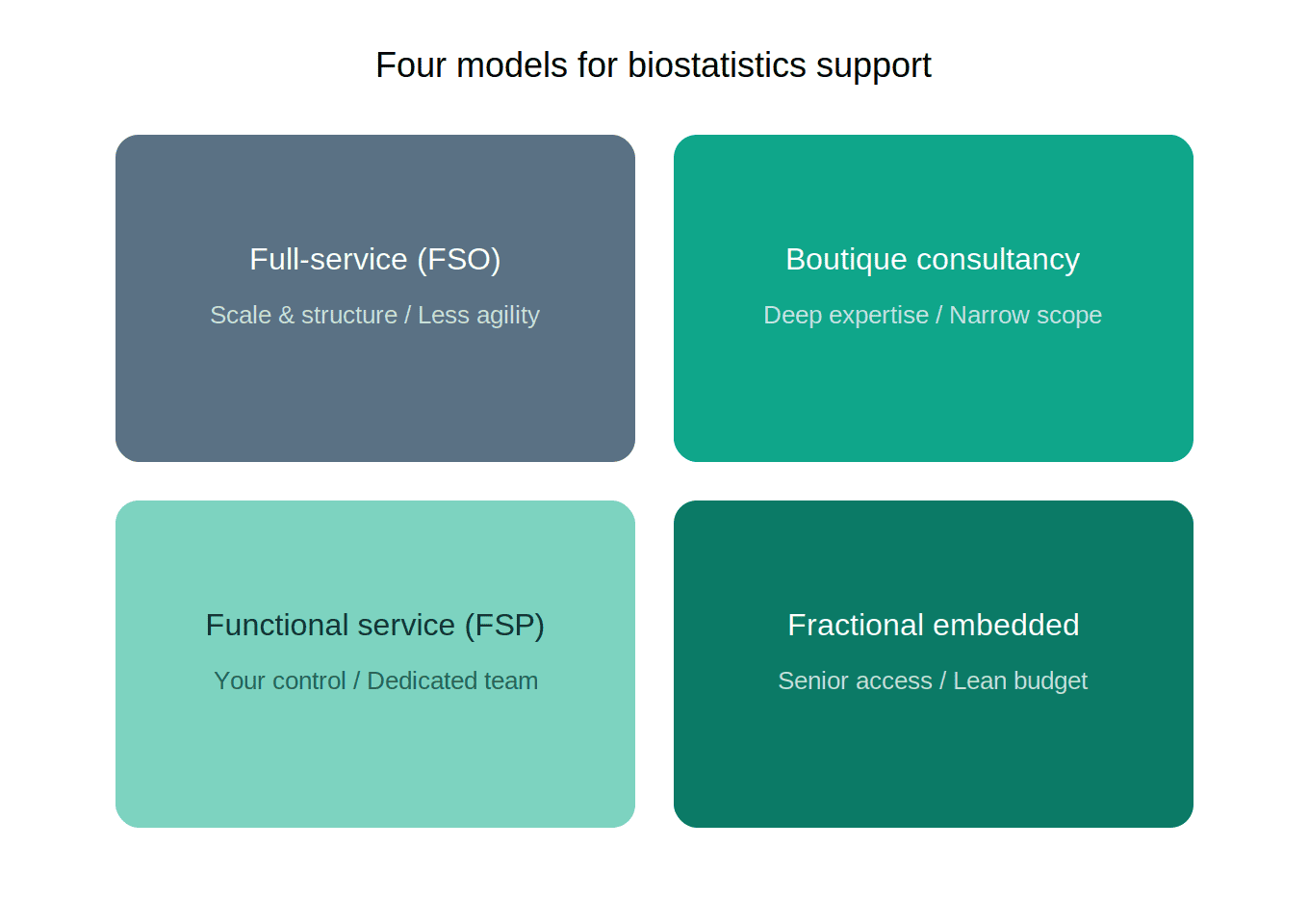
When outsourcing your biostatistics, you are choosing between four fundamentally different operational structures. Each carries distinct trade offs regarding cost, control, and depth of expertise.
1. Full Service Outsourcing (FSO) This is the traditional global CRO model (e.g., IQVIA, Labcorp). They manage your entire trial from site activation to statistical reporting.
Works when: You have a straightforward Phase III trial with standard endpoints, an massive budget, and no need for rapid clinical pivots.
Fails when: You are running adaptive designs, need strategic input on dose optimization, or require senior statisticians who understand your specific tumor type. Large CROs are built for scale, not agility.
2. Functional Service Provision (FSP) You outsource specific functions like biostatistics and programming while retaining overall project control.
Works when: You want dedicated statistical support integrated into your internal team to ensure continuity across multiple studies.
Fails when: You are a virtual biotech that lacks the internal infrastructure to manage the rest of the trial operations.
3. Boutique Consultancies Firms that focus entirely on complex trial design, adaptive protocols, and Bayesian modeling.
Works when: You are designing a complex BOIN dose escalation, a basket trial, or anything requiring advanced statistical simulation before enrollment begins.
Fails when: You need ongoing, day to day operational biostatistics support throughout trial execution.
4. Fractional or Embedded Model You engage a senior biostatistician on a part time basis to operate as a member of your leadership team. They direct trial design, manage downstream CRO vendors, and represent your statistical strategy in FDA meetings.
Works when: You are a lean biotech that needs top tier strategic statistical leadership without the cost of a full time executive hire.
Fails when: You have continuous, high volume programming needs that require a dedicated headcount.
What Biostatistics Actually Costs
Pricing variance in this space is enormous and depends heavily on the expertise level and engagement structure.
Academic collaborators: $135 to $250 per hour for PhD level statisticians.
Commercial subject matter experts: $250 to $350+ per hour for senior consultants with proven regulatory track records in oncology adaptive designs.
Block pricing examples:
Basic support (summary statistics, power estimation): ~$500 for 4 to 8 hours.
Standard support (multivariable regression, propensity matching): ~$950 for 8 to 20 hours.
Advanced design (prospective RCT development, adaptive protocols): $2,500+ for 20 to 40 hours.
Within a typical oncology trial budget, biostatistics and data management together represent just 5% to 10% of total costs. However, this modest percentage carries disproportionate strategic weight. A flawless site activation budget is worthless if the statistical design is flawed.
The blended rate trap: Large CROs frequently quote highly attractive blended rates to win your business, only to push the actual programming work to offshore junior staff. Always ask exactly who will personally perform your statistical analysis, not just who will oversee the account.
The Pain Points That Kill Programs
The Bait and Switch
This is the most frequent complaint from early stage biotechs. During the proposal phase, the CRO brings their most senior oncology biostatisticians to the table. After the contract is signed, your project is handed to a fragmented, junior team that lacks the context to identify statistical anomalies in real time.
The Fix: Write named personnel into your contract with replacement approval rights. Demand that the lead biostatistician from the pitch remains actively assigned to your project.
Protocol Amendments
Amendments are where clinical budgets go to die. The average direct cost of a single protocol amendment reaches $160,000 to $350,000, and that does not include the financial burn of timeline delays.
The cost breakdown of a typical amendment:
IRB/EC resubmission: $20,000 to $50,000
Site and vendor retraining: $50,000 to $100,000
EDC reprogramming: $25,000 to $75,000
Subject re-consenting: $10,000 to $25,000
Project team overhead: $50,000 to $100,000
The vast majority of these amendments are entirely avoidable. They stem from inadequate sample size estimation or poorly defined endpoints that were not properly vetted. A qualified biostatistics partner catches these issues during protocol development, long before enrollment begins.
Communication Failures
Industry data shows that 86% of sponsors cite poor communication as the ultimate red flag in failing CRO relationships. If you are experiencing delayed data cleaning with no operational explanation, or if your partner cannot articulate how specific statistical methods support your overall regulatory strategy, the relationship is already failing.
5 Questions That Separate Experts from Pretenders
When evaluating a biostatistics partner, you have to move beyond checking resumes. These questions expose whether they have true applied expertise.
1. How do you handle missing data and informative censoring within the estimand framework?
Why it matters: Missing data is inevitable in oncology trials. The candidate must be able to distinguish between random missing data and informative censoring (e.g., a patient dropping out due to toxicity versus simply relocating).
Red flag: Generic responses about standard imputation methods that completely ignore the clinical context.
2. Describe your experience designing trials aligned with FDA Project Optimus mandates.
Why it matters: Any reliance on legacy 3+3 dose escalation is now disqualifying for modern targeted therapies. They must be able to discuss randomized parallel dose response evaluation and PK/PD integration.
Red flag: Unfamiliarity with Project Optimus or an inability to explain how it fundamentally alters Phase I design.
3. What software do your teams use for adaptive trial simulations?
Why it matters: SAS and R proficiency is just the baseline. For advanced oncology adaptive designs, the candidate needs experience with specialized simulation software like FACTS or EAST Bayes.
Red flag: Zero simulation experience. Adaptive designs require modeling thousands of trial permutations before a single patient is enrolled.
4. How do you establish SOPs for peer review and independent data validation?
Why it matters: This reveals their quality management rigor. Regulatory rejections frequently trace back to simple data inconsistencies that a strict peer review process would have caught.
Red flag: Vague references to internal quality processes without any specific procedural details.
5. Can you walk me through a trial where you navigated divergent FDA and EMA requirements?
Why it matters: Global oncology programs require simultaneous regulatory alignment. The candidate should explain how they structure the Statistical Analysis Plan (SAP) to satisfy both agencies using one estimand strategy as the primary and another as a sensitivity analysis.
Red flag: Experience strictly limited to single region submissions.
Red Flags That Should End the Conversation
Beyond their interview answers, watch for these structural warning signs:
High staff turnover: Frequent personnel changes indicate operational instability. Losing your lead biostatistician mid trial is devastating.
No direct FDA interaction experience: Biostatisticians play a central role in Type B and Type C meetings. If your partner has never defended a design to FDA statisticians, they lack critical context.
Generic protocol templates: Oncology trials require hyper specific statistical design. A partner offering a cookie cutter template for solid tumors does not understand the nuances between different indications.
The Regulatory Landscape You Must Navigate
Project Optimus Is Not Optional
The FDA now mandates that sponsors move beyond the Maximum Tolerated Dose (MTD) toward precision dosing that maximizes efficacy while minimizing toxicity. Recent guidance explicitly requires randomized evaluation of benefit risk across a dose range before pivotal trials begin.
Your biostatistics partner must integrate Bayesian adaptive modeling and parallel dose response studies into your Phase I design. If they do not understand this, your clinical program will face an immediate clinical hold.
FDA vs EMA: The Divergences That Matter
Both agencies cooperate through ICH harmonization, but their approaches differ meaningfully when it comes to oncology.
The FDA has a higher tolerance for uncertainty. They are willing to accept surrogate endpoints and accelerated pathways, but they require raw data in CDISC format to conduct independent statistical re-analysis. The EMA is more conservative. They emphasize mature overall survival data and long term safety, relying heavily on sponsor analyzed reports rather than raw data re-analysis.
The Refuse to File (RTF) Risk
An FDA Refuse to File action stalls your application within 60 days of submission. In oncology, RTF decisions almost always trace back to statistical deficiencies such as invalid noninferiority testing margins, severe randomization imbalances, or unaddressed informative censoring biases.
The only mitigation is early regulatory engagement. Your biostatistics partner should be the one actively framing the statistical narrative for your FDA meetings.
The Case for True Specialization
The clinical landscape is littered with biologically active compounds that failed purely due to statistical errors.
For example, an FDA review of 22 case studies where Phase II results failed to replicate in Phase III found that failures occurred even with large Phase II trials. The common thread was unvalidated surrogate endpoints, inadequate sample size powering, and a failure to account for population variance.
Conversely, biotechs that engage highly specialized, embedded biostatistics support achieve vastly superior outcomes. Lean, high trust relationships with specialized teams enable rapid decision making without the bureaucratic drag of a massive CRO.
The Decision Framework
For early stage oncology biotechs, the biostatistics partner decision comes down to three final questions:
Does this partner have demonstrable, oncology specific expertise (survival analysis, competing risks, adaptive dose finding)?
Can this partner navigate both FDA and EMA estimand requirements simultaneously?
Will I have direct access to senior expertise, or will my project be delegated to a junior team after the contract is signed?
The premium for specialized biostatistics expertise is real, but it is a fraction of the cost of a single protocol amendment, a clinical trial rescue, or a Refuse to File action. Choose accordingly.
Our Recent Blog

Your cancer trial data deserves more than a generalist.
Whether you need a statistical analysis plan for your Phase II solid tumor study or a network meta analysis for your NICE submission, let's talk.

Your cancer trial data deserves more than a generalist.
Whether you need a statistical analysis plan for your Phase II solid tumor study or a network meta analysis for your NICE submission, let's talk.

Your cancer trial data deserves more than a generalist.
Whether you need a statistical analysis plan for your Phase II solid tumor study or a network meta analysis for your NICE submission, let's talk.

